Blockchain start-up Chronicled, which links the physical and digital worlds with cryptographic identity chips and blockchain technology and recently raised $6.25M, and the LinkLab, a life sciences supply chain consultancy, have launched a serialized ‘track and trace’ pilot for the pharmaceutical industry.
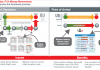
The pilot launched at a one-day event in San Francisco, where the start-up gathered feedback on the launch of a blockchain-based compliance protocol for that will satisfy the Drug Supply Chain Security Act. The goal of the pilot is to build an electronic, interoperable system to identify and track prescription drugs as they are distributed in the United States. It has come as a response to requirements from US FDA to better protect consumers by improving counterfeit detection and removal of potentially dangerous medicines.
Chronicled and the LinkLab have partnered to bring the protocol and compliance solution to market. This solution aims to provide the pharmaceutical industry with a less expensive, less cumbersome, and more secure approach to meeting existing regulatory requirements. The project’s goal is to develop around GS1 standards and to emphasize data privacy.
Susanne Somerville, co-founder of the LinkLab, commented: “The first phase of this project is to prove that one global pharmaceutical manufacturer can comply with their DSCSA regulatory obligations and meet the 2017 and 2023 requirements,” we are excited to be partnering with a major pharmaceutical player in this first phase.”
Upcoming phases will involve multiple players in the pharma supply chain, from manufacturer to pharmacy and hospitals, in order to establish a minimum viable ecosystem. In the future, these pilots may allow the industry to automate the flow of goods, finances, and documents leveraging smart contracts.
Samantha Radocchia, CPO and Co-founder at Chronicled, said: “We will be working closely with teams at leading enterprise blockchain projects over the coming months to identify the most suitable enterprise blockchain to serve as a data utility for the pharmaceutical industry,”